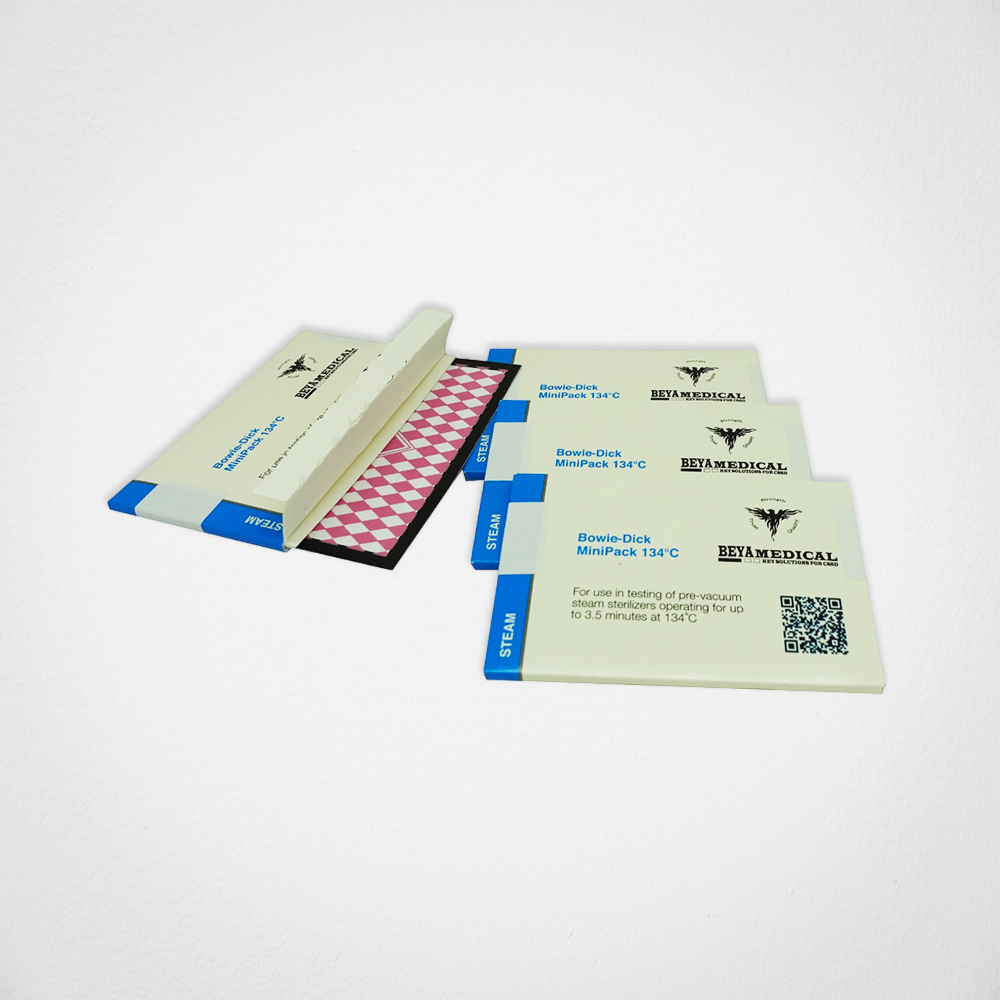
Bowie Dick Mini Pack
Bowie Dick Mini Pack 134 C test kit is intended for the daily monitoring of pre-vacuum steam sterilizers operating at 134 C for 3.5 minutes Compared to standard Bowie-Dick test packages, you can make savings in the form of less paper waste and space requirements for storage as well as lower transport costs.
When should a Bowie Dick Mini Pack be performed?
The Bowie Dick Mini Pack should be performed daily, whenever the sterilizer is in use, and before the first processed load. If your sterilizers remain on continuously, you should perform the test at the same time each day. The test pack is packaged in an easy to open box and designed to operate in sterilizers with sub-atmospheric or trans-atmospheric cycles as defined in EN ISO 11140-4.
What is history of Bowie Dick Test ?
The Bowie Dick test, name comes from their developers, J. H. Bowie and J. Dick, was first described in a 1963 publication titled The Bowie and Dick Autoclave Tape Test. At the time, it was known that the presence of a important amount of residual air in the sterilizer chamber of vacuum sterilizers, caused by an air leak or inadequate vacuum, would prevent thorough steam penetration into the load contents during the steam exposure phase of the sterilization cycle. However, a method for verifying the proper operation of vacuum sterilizers was not available. The paper by J. H. Bowie and his colleagues explained a quick and reliable method for verifying the operation of the vacuum system of vacuum sterilizers.
Which sterilization process is Bowie Dick Mini Pack designed for ?
Bowie Dick Mini Packs are designed to be used in Steam sterilizers.
Which sterilization cycle are Bowie Dick Mini Packs working with ?
Bowie Dick Mini Pack are designed operate at steam cycle 134 C – 3,5 minutes or 121 C – 15 minutes conditions.
What is the standart of ISO for Bowie Dick Mini Pack ?
Bowie Dick Mini Pack according to ISO 11140-1, ISO 11140-3 and ISO 11140-4 standart. CE marking is not applicable for indicator products, because this product is not considered a medical device under the clause of the EC-directive.